Zithrocan
August 21, 2024
Imigran
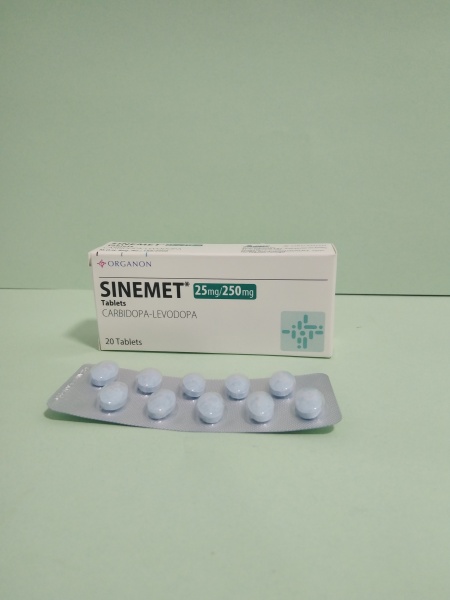
August 26, 2024Sinemet
90.00 EGP
25mg/250mg tablet
Tradename:
Sinemet
Compound:
Each tablet contains:
Carbidopa 25 mg
Levodopa 250 mg.
Auxiliary components:
Indigotine, corn starch, microcrystalline cellulose, magnesium stearate.
Properties:
Antiparkinsonian drug (combination of a dopamine precursor and a peripheral decarboxylase inhibitor).
Indications:
Parkinson’s disease and parkinsonism syndrome of known etiology (due to encephalitis, cerebrovascular disorders, intoxication with toxic substances, including carbon monoxide or manganese).
Directions for use and dosage:
Inside, with a small amount of food or after a meal, with water and without chewing. Since there is competition between aromatic amino acids and levodopa for absorption, large amounts of protein should be avoided while using the drug. The average daily dose of carbidopa required to suppress the peripheral conversion of levodopa is 70-100 mg. Exceeding 200 mg of carbidopa does not entail a further increase in the therapeutic effect. The daily dose of levodopa should not exceed 2000 mg. The initial dose is 1/2 tablet. 2 times a day, if necessary, can be increased by 1/2 tablet. /day As a rule, at the beginning of replacement therapy, the daily dose should not exceed 3 tablets. /day (1 tablet 3 times/day). Use at this dosage is recommended at the beginning of treatment of severe cases of parkinsonism. The daily dose of the drug, as an exception, can be increased during monotherapy, but should not exceed 8 tablets. (1 tablet 8 times/day). Use in quantities of more than 6 tablets. /day should be carried out with great care.
Contraindications:
Angle-closure form of glaucoma;
Severe psychosis or neurosis;
Melanoma or suspicion of it;
Skin diseases of unknown etiology;
Huntington’s disease;
Essential tremor;
Simultaneous use of non-selective MAO inhibitors, an interval of less than 2 weeks after the end of taking MAO inhibitors;
Pregnancy;
Lactation;
Hypersensitivity to the components of the drug.
Precautionary measures:
Sudden discontinuation of levodopa is unacceptable.
Monitoring of patients who need to suddenly reduce the dose of the drug or interrupt its use is necessary, especially if the patient is receiving antipsychotic drugs.
This combination is not used to eliminate extrapyramidal reactions caused by drugs.
During the treatment process, it is necessary to monitor the patient’s mental status and peripheral blood picture.
High protein foods may interfere with absorption.
Patients with glaucoma while taking this combination should regularly monitor intraocular pressure.
During long-term treatment, periodic monitoring of the functions of the liver, hematopoiesis, kidneys and cardiovascular system is advisable.
Before planned general anesthesia, treatment with this combination is continued as long as the patient is allowed to take it orally. After surgery, the usual dose can be restarted as soon as the patient is able to take the drugs orally.
Side effects:
From the cardiovascular system: arrhythmia and/or palpitations, orthostatic reactions, including a decrease or increase in blood pressure, fainting; phlebitis.
From the digestive system: vomiting, anorexia, diarrhea, constipation, dyspepsia, dry oral mucosa, change in taste, darkening of saliva, bleeding from the gastrointestinal tract, duodenal ulcer.
From the hematopoietic system: leukopenia, thrombocytopenia, anemia, including hemolytic, agranulocytosis.
From the nervous system: dizziness, headache, drowsiness, NMS, episodes of bradykinesia (on-off syndrome), sleep disturbances, including nightmares, insomnia; psychotic reactions, including delusions, hallucinations and paranoid thinking, confusion, agitation, paresthesia, depression (including suicidal thoughts), dementia, increased libido.
Allergic reactions: angioedema, urticaria, itching, hemorrhagic vasculitis (Henoch-Schönlein purpura), bullous rashes (including reactions similar to pemphigus).
From the respiratory system: shortness of breath, upper respiratory tract infections.
From the skin: skin rash, increased sweating, darkening of sweat, alopecia.
From the urinary system: urinary tract infections, frequent urination, dark urine.
From the laboratory parameters: decreased hemoglobin and hematocrit, increased activity of ALT, AST, LDH, alkaline phosphatase, hyperbilirubinemia, increased urea nitrogen, positive Coombs test, hyperglycemia, leukocyturia, bacteriuria and hematuria.
Other: chest pain, asthenia.
Adverse reactions that were observed with the use of levodopa alone can therefore be observed when using a combination of levodopa and carbidopa: myocardial infarction, abdominal pain, dysphagia, drooling, flatulence, bruxism, burning sensation of the tongue, heartburn, hiccups, edema, weight loss or increase. , ataxia, extrapyramidal disorders, falls